The chemistry of making PVC involves taking the simplest units, called monomers, and linking these monomer molecules together in a polymerization process to form long molecular chains called polymers (also called macromolecules).
This is the case of PVC, which is made from vinyl chloride monomer, usually known by its initial VCM through polymerization. Some monomers exist as reactive gaseous chemicals and some of these monomers can cause health hazards when in direct contact with humans. In these cases, they are produced and processed under strict controls to protect health, safety and the environment.
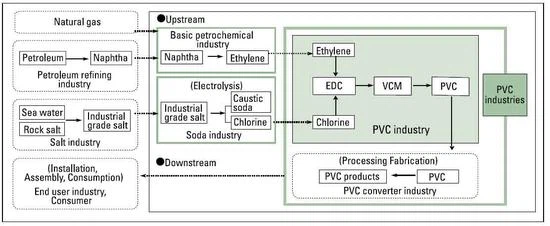
On the other hand, polymers such as PVC are made from monomers by the polymerization and are solid and chemically stable substances and therefore do not affect human health.VCM is the raw material of PVC and is a gas at ambient temperature, but is usually stored as a liquid under pressure. Ethylene and chlorine are the raw materials for PVC.
Through the thermal cracking of naphtha or natural gas, the basic petrochemical industry produces ethylene and propylene, among others. Naphtha is mainly supplied by the petroleum refining industry, which uses crude oil as a feedstock. The Chlor-alkali industry produces caustic soda, chlorine and hydrogen by electrolysis, using industrial-grade salt as the main raw material.
In the first stage of the PVC production process, ethylene and chlorine are mixed to produce an intermediate product called dichloroethane; this is then converted into vinyl chloride, the basic building block of polyvinyl chloride or PVC. The "polymerization" process joins the vinyl chloride molecules together to form PVC chains. The PVC produced in this way is in the form of a white powder. This is not used alone but is mixed with other components to provide formulations for a variety of products.
Most commercial plastics have carbon and hydrogen as their main components. PVC differs in that it contains chlorine (about 57% by weight) as well as carbon and hydrogen. The presence of chlorine in the molecule makes PVC particularly versatile, as it makes it compatible with a variety of other materials. The chlorine content also helps to make PVC flame retardant. It can also be used as a "marker" to distinguish PVC in automated sorting systems for plastic recycling. PVC formulations can be molded by a variety of techniques and use very little energy to make the final product form. PVC polymers are chemically stable, neutral and non-toxic. such as medical devices, as well as construction, automotive and cable.




